Commercial Summaries
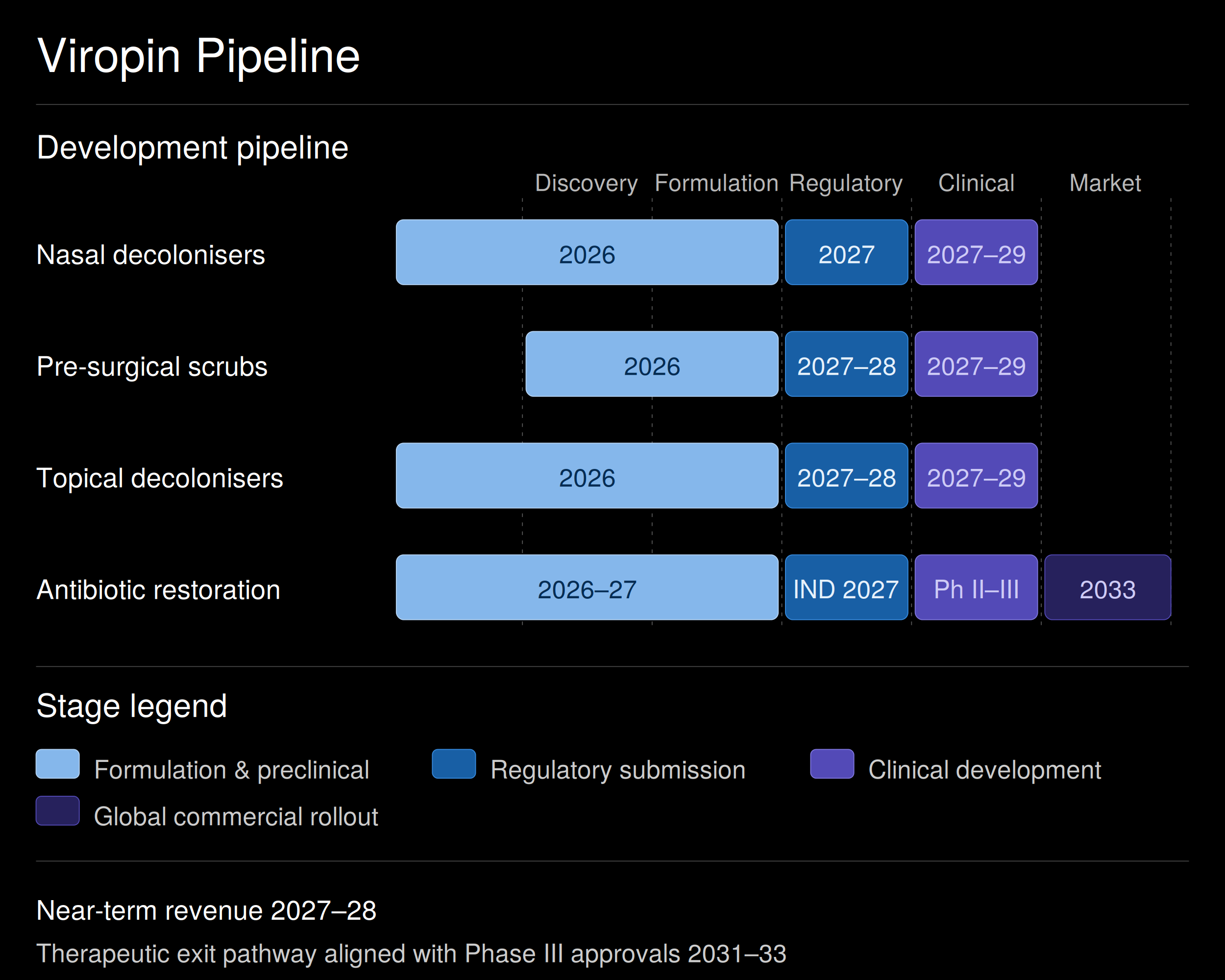
MRSA Nasal & Topical Decolonisation
For hospitals managing MRSA-carrier patients before admission, surgery, or transfer, aged care, and community health programmes. MRSA is one of the most costly and persistent hospital pathogens globally. Effective decolonisation directly reduces infection rates, length of stay, and antibiotic use.
Clinical development 2027–2029, with potential fast-track regulatory status in key markets, given the public health priority. International commercial partnerships under development.
Pre-Surgical Scrubs
Designed for surgical units, intensive care, and aged-care facilities. Hospital procurement teams already budget for this category. Viropin's products are formulated to outperform on efficacy against resistant organisms while integrating into existing infection-control protocols. Standard antiseptic and infection-control regulatory pathways across major markets.
Formulation development underway through 2026; regulatory submissions planned across major markets 2027–2028.
Hospital Decolonisers & Antiseptics
Targeted at hospital networks, infection-control teams, and aged-care providers. Used for environmental sanitation and patient decolonisation across the patient journey. Supporting protocols that reduce the spread of resistant bacteria and lower downstream antibiotic use.
Same regulatory pathway and timeline as the pre-surgical scrubs line, allowing parallel development and a shared commercial launch.
Antibiotic Restoration Therapy
Addressing the $157B antimicrobial resistance market growing at 15.3% CAGR. Designed for global hospital and clinical use against the highest-priority resistant pathogens on WHO and CDC watchlists. Restoring existing antibiotics is faster, cheaper, and clinically more deployable than developing new ones. Preclinical mechanistic testing, MIC analysis, and resistance modelling are underway with a leading CRO. IND submission and Phase II initiation 2027–2028, global Phase III 2029–2031, regulatory submissions across the US, Europe, the Middle East and Asia 2031–2032. Letter of Intent signed with major international pharma.
IPO or strategic exit aligned with late-stage approvals.
